FDA plans ultra-fast review of three psychedelic drugs following Trump directive
The Food and Drug Administration says it will offer ultra-fast review to three psychedelic drugs being studied for hard-to-treat mental health conditions, including major depression
April 24, 2026
 US-Mexico border wall construction is desecrating sacred sites, Indigenous leaders say
US-Mexico border wall construction is desecrating sacred sites, Indigenous leaders say
 Palestinian leader's son wins role in Abbas' party, official says
Palestinian leader's son wins role in Abbas' party, official says
 Community college student from Michigan to be 1st woman to represent US at world welding competition
Community college student from Michigan to be 1st woman to represent US at world welding competition
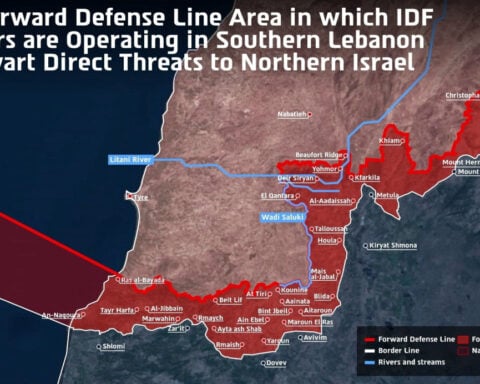
 Israel Q1 GDP shrinks 3.3% annualised as Iran war weighs
Israel Q1 GDP shrinks 3.3% annualised as Iran war weighs
 3 Finnish divers arrive in the Maldives to remap the search for the bodies of 4 Italian divers
3 Finnish divers arrive in the Maldives to remap the search for the bodies of 4 Italian divers
 Demand for cruises appears undimmed despite hantavirus and other onboard outbreaks
Demand for cruises appears undimmed despite hantavirus and other onboard outbreaks